What to know about the first drug to treat fatty liver disease
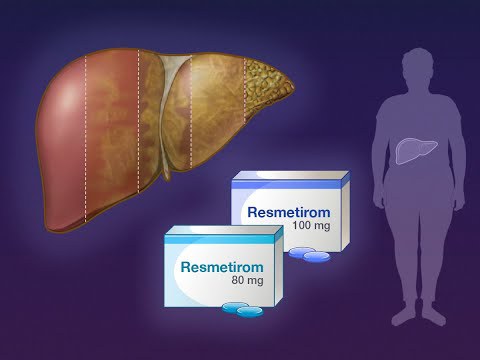
By A.J. Hostetler
The U.S. Food and Drug Administration has approved the first drug to treat fatty liver disease, which affects as many as 15 million adults in the United States.
The approval is “a major milestone for our patients and for the field,” according to Arun Sanyal, M.D., director of the VCU Stravitz-Sanyal Liver Institute for Liver Disease and Metabolic Health and an investigator in the clinical trial for the drug resmetirom, now branded as Rezdiffra.
Fatty liver disease, also known as steatotic liver disease, causes the body to begin storing excess fat in the liver. This doesn’t always cause problems for people, but when it does, the buildup of fat can cause inflammation and damage. The advanced stage of this is called metabolic dysfunction-associated steatohepatitis, or MASH, which is the leading cause of liver-rated medical conditions and mortality.
“With approved resmetirom, our patients with clinically significant scarring of the liver due to MASH will have access to an effective and approved therapeutic agent to reverse disease progression,” Sanyal said. “I congratulate the communities of scientists and patients who participated in the treatment trials to make approved therapy for MASH a reality.”
Rezdiffra is expected to be available for patients starting in April 2024. Sanyal spoke with VCU Health News to help patients, caregivers and health care providers better understand this new drug and how it may change the future of liver treatment.
What medical condition does Rezdiffra treat?
Rezdiffra treats a condition called metabolic dysfunction-associated steatohepatitis or MASH. MASH occurs when fat accumulates in the liver and causes inflammation. As it worsens, it can cause scarring, known as fibrosis, and can continue to advance to significant fibrosis or even cirrhosis, cancer, and the need for a liver transplant.
Who is the drug intended to treat?
MASH affects approximately 6 to 8 million people in the United States. It is often associated with other health problems such as high blood pressure, type 2 diabetes, obesity and high blood fat levels. The newly approved medication is intended for people with MASH with moderate to advanced fibrosis – when patients are said to be on the cusp of cirrhosis.
There are other drugs undergoing clinical testing to treat patients whose fibrosis is less advanced and could possibly halt the scarring or even lessen the amount of fat in the patient’s liver.
How does Rezdiffra work?
Until now, physicians treating MASH patients focused on managing the disease through weight loss from lifestyle changes such as a healthy diet and regular exercise.
The newly approved medication, Rezdiffra, targets a protein directly in the liver and reduces liver fat, inflammation, and scarring. Clinical trials found it can halt the progression of fibrosis or even reverse it. It’s intended to be used along with a healthy diet and exercise.
Are there any safety concerns or side effects to this treatment?
Patients should talk to their health care provider about whether this medication suits their situation. They should not have to undergo a potentially risky liver biopsy for diagnosis to be prescribed Rezdiffra, but those with MASH and stage 2 or 3 fibrosis can be identified with noninvasive tests.
The drug is taken by mouth. Its most common side effects include diarrhea and nausea. Rezdiffra comes with certain warnings and precautions, such as drug-induced liver toxicity and gallbladder-related side effects. Patients with a severe form of cirrhosis, called decompensated cirrhosis, should avoid the drug. Patients using Rezdiffra who develop signs or symptoms of worsening liver function should stop taking it and talk to their health care provider.
Why is the approval of this fatty liver disease treatment so significant?
Liver disease is often called “silent” because so many patients don’t feel any symptoms and their illness goes undiagnosed, but it is the ninth leading cause of death in the U.S. and is an increasing burden on health care systems globally. Patients with MASH, especially those with metabolic diseases like obesity or type 2 diabetes, are at greater risk for adverse cardiovascular events and increased illness and mortality.
The problem is so pressing that the FDA approved Rezdiffra under its accelerated approval pathway, which allows for earlier approval of drugs that treat serious conditions and address an unmet medical need. Madrigal Pharmaceuticals, the company that created Rezdiffra, must complete a post-approval study to verify the drug’s clinical benefit, which was outlined in a recent article in the New England Journal of Medicine.
link