Middle East Liver Cancer Diagnostics Market Report, 2033
Market Size & Trends
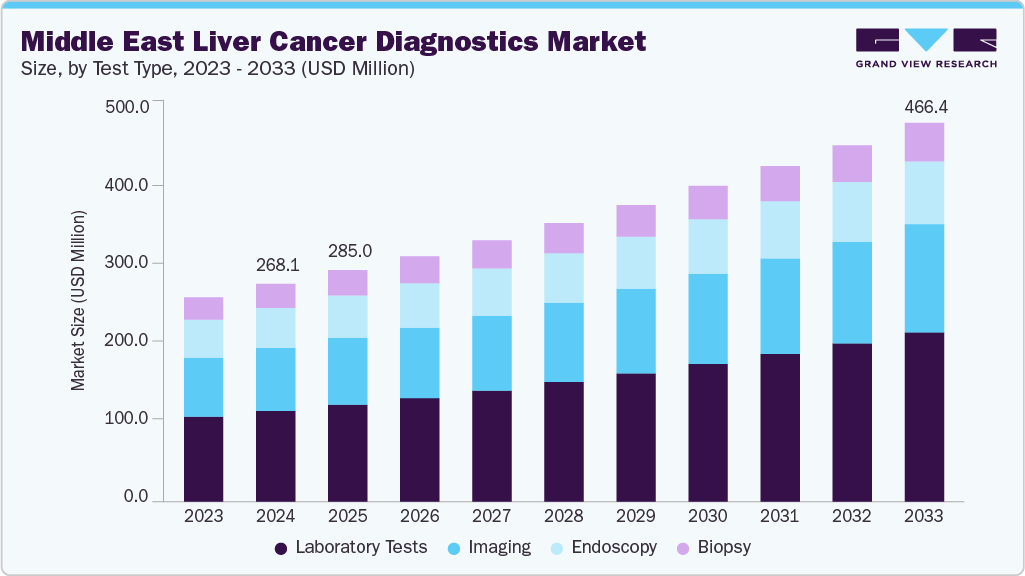
The Middle East liver cancer diagnostics market size was estimated at USD 268.10 million in 2024 and is projected to reach USD 466.42 million by 2033, growing at a CAGR of 6.35% from 2025 to 2033. The ME liver cancer diagnostics market is primarily driven by the high prevalence of HBV and HCV, with Egypt reporting some of the region’s highest infection burdens. Rising cases of metabolic dysfunction-associated fatty liver disease (MAFLD) and diabetes are accelerating incidence rates, particularly in Gulf countries. Despite progress in viral hepatitis treatment, chronic infection remains a key driver of hepatocellular carcinoma. Increasing investments in advanced diagnostic technologies, including liquid biopsy, NGS, and AI-driven platforms, along with national cancer control programs and improved registries, are further fueling market growth by supporting early detection and better patient outcomes.

The Middle East liver cancer diagnostics market is expanding rapidly, driven by epidemiological trends, strategic alliances, and accelerated adoption of advanced technologies. Hepatocellular carcinoma (HCC), the most common form of liver cancer, continues to rise across the region, with different etiological patterns shaping country-level dynamics. Egypt has long borne the burden of hepatitis C-driven liver cancer, while Gulf states such as Saudi Arabia, the UAE, Kuwait, and Qatar are witnessing increasing cases linked to obesity, diabetes, and metabolic syndrome. This epidemiological transition underscores the pressing need for sensitive, non-invasive diagnostic tools, supported by public health initiatives and stronger cancer registries. Despite progress in viral hepatitis management, lifestyle-related drivers continue to fuel incidence, requiring diagnostics that can enable earlier detection and intervention.
The competitive landscape is being reshaped by global biotech firms and regional healthcare leaders. In October, 2024, Helio Genomics entered a strategic partnership with Halub Medical Co. to commercialize HelioLiver Dx in Saudi Arabia and the broader GCC. The test, expected to be launched commercially in Q1 2025, is projected to generate more than USD 10 million in revenue in its first year of availability. HelioLiver Dx leverages AI-driven multi-analyte technology-analyzing cfDNA, methylation signatures, serum proteins, and demographic markers and demonstrated four-times higher sensitivity than ultrasound in pivotal U.S. trials led by Stanford Medical Centers. Importantly, it offers adherence rates nearly nine-times greater than conventional surveillance, addressing a critical gap in patient compliance across the Middle East. The partnership aligns with Saudi Arabia’s Health Cluster initiatives and Vision 2030 strategy, reflecting how international innovation is being localized to address regional healthcare burdens.
Earlier, CureLab Oncology established operations in the GCC in October 2020, emphasizing DNA-based therapeutics for cancer and chronic inflammatory conditions. By collaborating with hospitals and universities across Saudi Arabia, Kuwait, the UAE, Qatar, and Oman, CureLab created opportunities for local participation in international clinical trials while positioning the region as a hub for early-stage biomedical innovation. This move not only reflected confidence in the GCC’s market potential but also illustrated how Middle Eastern healthcare ecosystems are becoming testing grounds for cutting-edge oncology solutions. Meanwhile, Cleveland Clinic Abu Dhabi’s Fatima bint Mubarak Center has set new standards in clinical oncology care. The facility introduced AI-enabled adaptive radiotherapy (Ethos), robotic chemotherapy preparation systems, and Y90 radioembolization therapy. These innovations shorten treatment times, improve safety, and enhance targeting precision, raising the level of oncology care available locally and reducing reliance on medical tourism for advanced diagnostics and therapies.
Complementing these systemic and infrastructural advances, individual healthcare providers are demonstrating the effectiveness of personalized, multi-modality oncology strategies. Dr. Sulaiman Al Habib Hospital in Dubai recently reported the successful treatment of a complex liver tumor in a 46-year-old patient, achieved through a three-step regimen that combined systemic immunotherapy, Y90 radioembolization, and advanced surgical resection. This case highlighted the hospital’s ability to deliver highly tailored oncology care using a multidisciplinary approach and underscored the growing availability of precision oncology expertise within the region. Such examples demonstrate how the Middle East is not only adopting but also adapting global best practices in liver cancer diagnostics and treatment. Together, these initiatives point to a market undergoing rapid transformation, where rising disease incidence, government-backed healthcare reforms, and private-sector innovation converge to drive growth in early detection solutions. With increasing investment in AI-enabled diagnostics, blood-based testing, and integrated oncology centers, the Middle East liver cancer diagnostics market is positioned to expand significantly, delivering both improved patient outcomes and robust commercial opportunities over the coming decade.
Market Concentration & Characteristics
The degree of innovation in the Middle East liver cancer diagnostics market is steadily advancing, driven by both international collaborations and regional investments in precision medicine. Countries such as Saudi Arabia and the UAE are at the forefront, integrating AI-driven multi-analyte blood tests, adaptive radiotherapy, and advanced molecular diagnostics into their healthcare systems. Strategic partnerships, like Helio Genomics with Halub Medical, are accelerating the adoption of non-invasive, highly sensitive screening tools for hepatocellular carcinoma. Furthermore, new oncology centers, such as the Fatima bint Mubarak Center in Abu Dhabi, are setting benchmarks in diagnostic innovation, fostering regional leadership in early cancer detection.
The level of M&A activities in the Middle East liver cancer diagnostics market remains moderate but is showing signs of acceleration as global biotech and diagnostic companies seek regional footholds. Strategic alliances and partnerships are more common than outright acquisitions, with companies like Helio Genomics partnering with Halub Medical to commercialize HelioLiver Dx across Saudi Arabia and the GCC. International players are leveraging local healthcare providers to expand access, while regional hospitals and cancer centers are collaborating with global innovators to introduce advanced diagnostic platforms. This partnership-driven model is gradually paving the way for potential consolidation and future M&A activity.
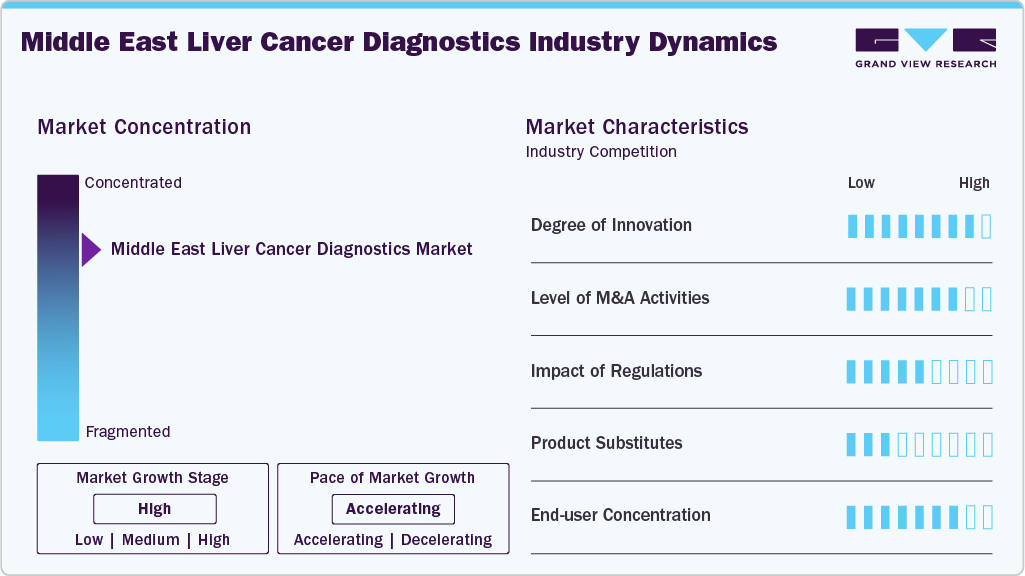
The impact of regulation on the Middle East liver cancer diagnostics market is significant, as each country operates under distinct healthcare frameworks that influence product approvals, reimbursement, and adoption timelines. Regulatory authorities such as the Saudi Food and Drug Authority (SFDA), UAE’s Ministry of Health and Prevention (MoHAP), and Qatar’s Ministry of Public Health (MOPH) are actively strengthening oncology diagnostic guidelines to align with global standards, emphasizing early detection and precision medicine. While approval pathways are becoming more structured, variations in data requirements, reimbursement policies, and clinical adoption across countries often delay uniform market entry. Nevertheless, ongoing reforms aimed at harmonization and faster approval of innovative diagnostics are creating a more favorable environment for advanced technologies.
In the Middle East liver cancer diagnostics market, product substitutes primarily include conventional diagnostic approaches such as ultrasound, CT scans, MRI, and alpha-fetoprotein (AFP) blood tests, which remain widely used due to availability and lower cost. However, these methods have limitations in sensitivity, particularly for detecting early-stage hepatocellular carcinoma (HCC). As a result, newer blood-based multi-analyte tests and liquid biopsy solutions are emerging as superior alternatives. While substitutes continue to dominate initial screening in public healthcare systems, the growing push for early detection and precision medicine is steadily shifting demand toward advanced molecular diagnostics, thereby reducing reliance on traditional tools.
In the Middle East liver cancer diagnostics market, end-user concentration is relatively high, with a few large tertiary care hospitals, specialized cancer centers, and government-backed healthcare clusters driving the majority of testing volumes. Institutions such as Cleveland Clinic Abu Dhabi, King Faisal Specialist Hospital in Saudi Arabia, and Dr. Sulaiman Al Habib Hospitals act as key hubs for advanced diagnostic adoption. Public hospitals dominate routine screening and surveillance programs, while private centers increasingly invest in cutting-edge blood-based and imaging diagnostics. This concentration reflects limited high-end diagnostic infrastructure across the region, creating strong influence of a few major end-users on overall market adoption.
Test type Insights
The laboratory tests segment dominated the liver cancer diagnostics market in 2024, accounting for the largest revenue share of 41.73% and expanding at a CAGR of 7.19%. This growth is fueled by the rising prevalence of liver cancer and the critical role of laboratory testing in screening high-risk patients, staging tumors, guiding treatment decisions, and monitoring disease recurrence. Laboratory tests, particularly blood-based and biomarker assays, remain integral for early detection and precision treatment planning.
The broader diagnostics landscape is also shaped by the integration of biomarkers and advanced imaging modalities such as ultrasound, CT, and MRI, which improve diagnostic accuracy when combined with laboratory tests. Endoscopy and biopsy remain vital for confirmation and staging, reinforcing a multi-modal approach to diagnosis.
Recent innovations further illustrate the segment’s momentum. In April 2025, Bayer and Siemens Healthineers launched advanced imaging systems, including the MEDRAD Centargo CT Injection System, MAGNETOM Flow MRI, and SOMATOM Pro.Pulse Dual Source CT scanner. These technologies enhance imaging speed, accuracy, and efficiency-offering AI-driven improvements and high-resolution capabilities that complement laboratory testing and strengthen diagnostic precision.
Together, the synergy between laboratory tests, biomarker innovations, and next-generation imaging platforms is transforming liver cancer diagnostics, enabling earlier intervention, more accurate disease characterization, and better patient outcomes.
End-use Insights
In 2024, hospitals and diagnostic laboratories led the liver cancer diagnostics market with a 49.26% share, underscoring their central role in early detection and patient management. Hospitals leverage advanced infrastructure and specialized oncology units to conduct imaging, biopsies, biomarker assays, and blood tests for comprehensive diagnosis, while diagnostic laboratories ensure precise sample analysis that informs treatment decisions. Rising liver cancer incidence has further amplified their significance, making these institutions indispensable for accurate and timely diagnosis.
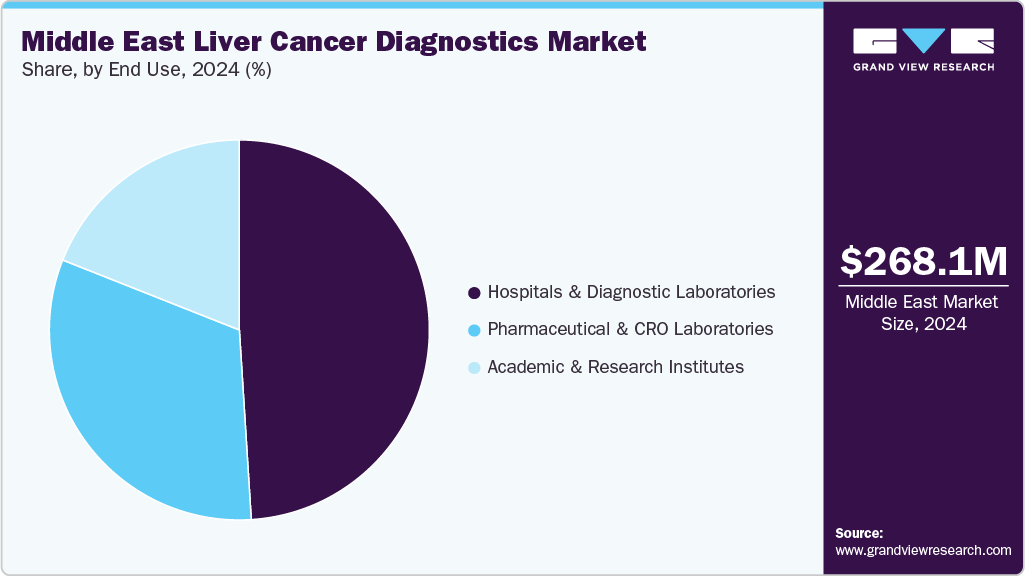
Pharmaceutical companies and CRO laboratories are also accelerating innovation in the liver cancer diagnostics and treatment ecosystem through drug launches and research collaborations. For instance, in June 2023, AstraZeneca announced the launch of IMJUDO (tremelimumab) in the UAE, making it one of the first Middle Eastern countries to gain access to this breakthrough cancer therapy. Clinical trials have demonstrated its efficacy in metastatic non-small cell lung cancer and hepatobiliary cancers, significantly improving survival outcomes. AstraZeneca introduced the therapy through workshops attended by 100+ healthcare professionals, highlighting the role of immunotherapy combinations in addressing high unmet needs in liver and lung cancer. This development reinforces the complementary role of pharmaceutical innovation alongside CRO-led research and diagnostics in advancing precision oncology solutions.
Country Insights
The Middle East liver cancer diagnostics landscape is accelerating, shaped by hepatitis B/C endemics, rising MAFLD/diabetes, and aging populations. Gulf markets (Saudi Arabia, UAE, Qatar, Kuwait, Oman) are scaling biomarker-led surveillance and imaging, while expanding NGS panels and liquid biopsy for earlier HCC detection. Saudi Arabia’s large public-hospital network and centralized procurement spur volume adoption; the UAE advances with AI-enabled imaging, Y-90 radioembolization programs, and blood-based surveillance pilots. Qatar’s precision-health infrastructure (biobanks, genome programs) strengthens population-tailored risk tools. Cross-border partnerships-industry, CROs, and hospital systems-are shortening validation cycles and improving access. Persistent gaps include low screening adherence and heterogeneous reimbursement, but policy momentum and clinical capacity are driving double-digit growth.
Saudi Arabia Liver Cancer Diagnostics Market Trends
The Saudi Arabia liver cancer diagnostics market is growing rapidly, driven by the government’s Vision 2030 initiative to modernize healthcare infrastructure. The growth of the country’s market can be largely attributed to the growing government involvement and heightened awareness of the advantages of non-invasive diagnostic methods. Moreover, significant advancements in NGS and PCR technologies have enhanced the accuracy of diagnostic procedures over time. Key drivers include increasing liver cancer prevalence, advancements in diagnostic technologies, and supportive government initiatives. The government’s focus on early detection and improved healthcare accessibility further contributes to the market’s expansion.
UAE Liver Cancer Diagnostics Market Trends
The UAE liver cancer diagnostics market is rapidly evolving, driven by rising hepatocellular carcinoma (HCC) incidence tied to metabolic syndrome, hepatitis prevalence, and diabetes. Hospitals and diagnostic labs dominate adoption, deploying NGS-based biomarker tests, liquid biopsy, and AI-enhanced imaging (e.g., adaptive radiotherapy, Y-90 radioembolization). Government initiatives-including the National Cancer Control Plan, expanded insurance coverage, and centralized procurement-are boosting testing infrastructure. Strategic partnerships, like HelioLiver Dx commercialization and collaborations in bioinformatics and lab networks, are expanding access and local capability. As surveillance demand increases, early detection tools are transitioning from niche to mainstream, signaling stronger market growth ahead.
Kuwait Liver Cancer Diagnostics Market Trends
The Kuwait liver cancer diagnostics market is witnessing steady growth, supported by rising liver cancer incidence linked to high obesity, diabetes, and metabolic-associated fatty liver disease (MAFLD) rates in the population. Hospitals remain the primary centers for diagnosis, leveraging imaging technologies such as CT, MRI, and ultrasound, alongside laboratory testing and biopsy for confirmation. Kuwait’s Ministry of Health has strengthened cancer screening and diagnostic infrastructure under national healthcare modernization programs, improving access to advanced tools. Growing collaborations with regional and international diagnostic providers, along with investments in precision medicine, are expected to accelerate adoption of innovative biomarker and molecular testing.
Qatar Liver Cancer Diagnostics Market Trends
The liver cancer diagnostics market in Qatar is witnessing steady progress, supported by government-led initiatives to improve cancer care infrastructure and early detection programs. With liver cancer ranking among the top health concerns in the region due to high prevalence of hepatitis and metabolic disorders, demand for advanced diagnostics such as imaging modalities, biomarker testing, and molecular diagnostics is rising. The country is investing in expanding laboratory and hospital capacities to strengthen precision medicine. Collaborations with international partners, alongside increasing adoption of digital health and AI-based imaging solutions, are further enhancing diagnostic accuracy and patient management in Qatar.
Oman Liver Cancer Diagnostics Market Trends
The liver cancer diagnostics market in Oman is gradually expanding, driven by the country’s growing focus on strengthening oncology services within its healthcare system. Rising incidence of liver cancer, primarily linked to viral hepatitis, obesity, and metabolic disorders, has heightened the demand for early detection tools. Hospitals and diagnostic laboratories are adopting advanced imaging techniques, biomarker assays, and molecular diagnostics to improve clinical outcomes. Government investment in cancer care infrastructure, along with initiatives to train healthcare professionals in precision diagnostics, is supporting growth. Additionally, collaborations with regional and global partners are enabling access to innovative diagnostic technologies in Oman.
Key Middle East Liver Cancer Diagnostics Company Insights
Some of the key players operating in the market include Abbott Laboratories, Thermo Fisher Scientific, Inc, F. Hoffmann-La Roche Ltd, Qiagen N.V, Siemens Healthineers, Becton, Dickinson & Company. New product launch, mergers and acquisitions, and collaborations are some of the major strategies adopted by these players to counter the stiff competition.
Key Middle East Liver Cancer Diagnostics Companies:
- Abbott Laboratories
- Thermo Fisher Scientific, Inc.
- F. Hoffmann-La Roche Ltd.
- Qiagen N.V.
- Siemens Healthineers
- Becton, Dickinson & Company
- Illumina, Inc.
- Epigenomics AG
- Koninklijke Philips N.V.
- Fujifilm Medical Systems U.S.A., Inc.
Recent Developments
-
In April 2022, the Viafet-Euformatics alliance introduced next-generation sequencing (NGS) assays and liquid biopsy platforms into the Middle East’s diagnostic ecosystem, marking a major step toward precision oncology. The collaboration combined Viafet’s regional expertise in clinical genomics with Euformatics’ advanced bioinformatics solutions, enabling laboratories to implement comprehensive hereditary and somatic mutation testing at scale. By introducing liquid biopsy technologies, the alliance also expanded access to minimally invasive cancer diagnostics, allowing physicians to track tumor progression and treatment response through circulating tumor DNA (ctDNA). This initiative not only addressed the growing demand for advanced hereditary and cancer screening tools in the region but also laid the groundwork for integrating AI-driven analytics and harmonized reporting standards, strengthening the foundation for precision medicine programs in the Middle East.
-
In February 2025, the Agilus-Lucence partnership expanded access to the LiquidHALLMARK test, a next-generation sequencing-based liquid biopsy that analyzes both circulating tumor DNA (ctDNA) and circulating tumor RNA (ctRNA) for biomarkers associated with multiple cancers, including liver malignancies. The initiative significantly broadened the regional availability of advanced molecular diagnostics by enabling hospitals and laboratories to incorporate liquid biopsy into routine workflows for early detection, treatment monitoring, and recurrence tracking. This collaboration underscored the growing role of precision diagnostics in oncology care, complementing global advancements. Notably, in January 2025, the FDA granted breakthrough device designation to EvoLiver, a novel test for hepatocellular carcinoma (HCC) surveillance in high-risk cirrhosis patients. Together, these milestones illustrate how regional institutions are aligning with international innovation trends to accelerate adoption of cutting-edge liver cancer diagnostic tools.
Middle East Liver Cancer Diagnostics Market Report Scope
|
Report Attribute
|
Details
|
|
Market size value in 2025
|
USD 285.04 million
|
|
Revenue forecast in 2033
|
USD 466.42 million
|
|
Growth Rate
|
CAGR of 6.35% from 2025 to 2033
|
|
Actual data
|
2021 – 2024
|
|
Forecast period
|
2025 – 2033
|
|
Quantitative units
|
Revenue in USD billion/million and CAGR from 2025 to 2033
|
|
Report coverage
|
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
|
|
Segments covered
|
Test type, end-use, country
|
|
Regional scope
|
Middle East
|
|
Country scope
|
Saudi Arabia; UAE; Kuwait; Oman; Qatar
|
|
Key companies profiled
|
Abbott Laboratories; Thermo Fisher Scientific, Inc.; F. Hoffmann-La Roche Ltd.; Qiagen N.V.; Siemens Healthineers; Becton; Dickinson & Company; Illumina, Inc.; Epigenomics AG; Koninklijke Philips N.V.; Fujifilm Medical Systems U.S.A., Inc.
|
|
Customization scope
|
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
|
|
Pricing and purchase options
|
Avail customized purchase options to meet your exact research needs. Explore purchase options
|
Middle East Liver Cancer Diagnostics Market Report Segmentation
This report forecasts revenue growth at regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the Middle East liver cancer diagnostics market report on the basis of test type, end-use, country:
-
Middle East Liver Cancer Diagnostics Test type Outlook (Revenue, USD Million, 2021 – 2033)
-
Laboratory Tests
-
Imaging
-
Endoscopy
-
Biopsy
-
-
Middle East Liver Cancer Diagnostics End-use Outlook (Revenue, USD Million, 2021 – 2033)
-
Hospitals & Diagnostic Laboratories
-
Academic & Research Institutes
-
Pharmaceutical & CRO Laboratories
-
-
Country Outlook (Revenue in USD Million, 2021 – 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Kuwait
-
Qatar
-
Oman
-
Rest of MEA
-
-
link